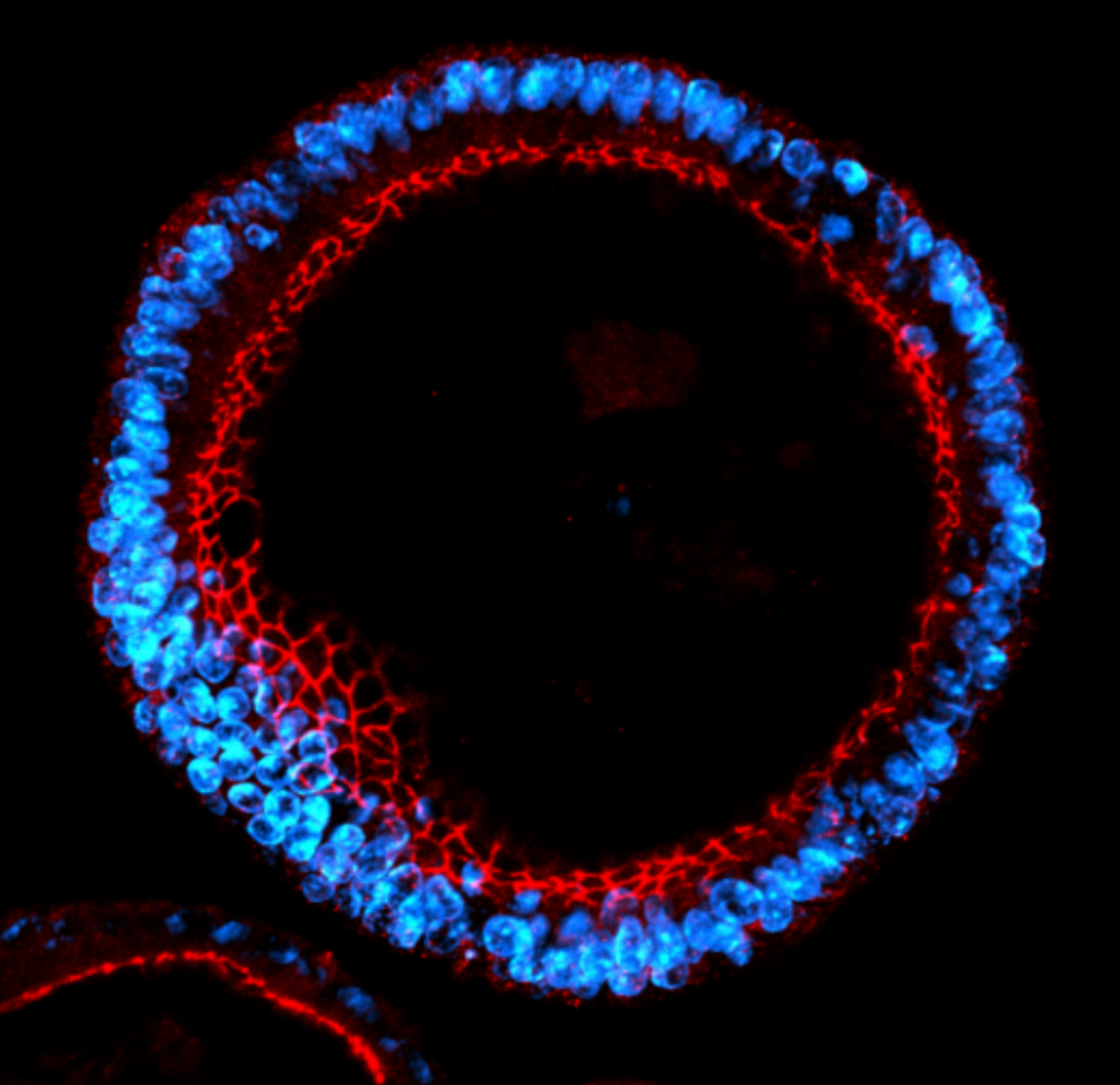
Confocal microscopy images of mouse colonic organoids stained for ZO-1 in red and cell nuclei in blue.
The CFIT Program provides both resources and training for CF researchers to pursue studies on CF patient-derived induced pluripotent stem cells (iPS cells) and other CF-relevant systems to advance the goal of Personalized Medicine in CF. Training courses are held in Toronto and are typically 3-5 days in length. Training travel awards of up to $ 2500 may be available for qualifying trainees.
Please watch this page for future training course offerings. If you are interested in a future offering of our lung differentiation workshop or a nasal cells workshop, please contact peckford@sickkids.ca to be placed on the list of interested candidates.
Details on a previous offering of the lung differentiation course are below:
Directed Differentiation of iPS Cells into Systems to Model CF Lung Disease
May 7-10th, 2019, Toronto, Canada
![]() Click here for the workshop poster
Click here for the workshop poster
This comprehensive 4-day workshop is on methods to maintain human iPSCs in culture, and directed differentiation of these cells to epithelial cells suitable as a model for lung disease and the study of Cystic Fibrosis. In this workshop trainees will have the opportunity to perform critical steps of the protocols. We will discuss and demonstrate important aspects of the workflow including characterization of patient-derived iPSCs and how they can be used as a model system for the study of CF lung disease. The workshop will include lectures by world renowned researchers involved in the generation and expansion of iPSCs from blood, and in the differentiation of these cells to lung epithelia and other CF-relevant model systems. This course is open to Canadian and international academic trainees. Limited space is available to industry participants. Workshop is presented by the CFIT Program in collaboration with the Ontario Institute for Regenerative Medicine.
Travel awards
There are three types of travel/registration cost reimbursement awards available. Trainees may apply to a maximum of one award program.
The CFIT Program is offering awards of up to $ 2500 for reimbursement of registration and travel expenses to individuals who do not reside in Toronto and who travel to attend the training course:
![]() Trainee travel award application form
Trainee travel award application form
OIRM Travel/registration awards
Ontario Institute for Regenerative Medicine (OIRM) is also offering travel/registration awards. These awards are only open to trainees in an OIRM investigator lab and will provide support for travel costs and registration support to a maximum of $ 1500, or for Toronto based trainees, registration fee support of $ 1200. For individuals from an OIRM lab outside of Toronto, use the CFIT travel award application form, however note that the maximum reimbursement is $ 1500 and the form should be submitted to jodi.garner@oirm.ca by March 31, 2019. For trainees from OIRM labs in Toronto, apply using the OIRM travel award application form.
![]() OIRM travel award application form (Toronto attendees)
OIRM travel award application form (Toronto attendees)
STEMCELL Technologies Travel Award
STEMCELL Technologies is a Canadian biotechnology company that develops specialty cell culture media, cell isolation systems, accessory products, and scientific services for life science research. As Scientists Helping Scientists, STEMCELL is committed to supporting training and mentorship for the scientific community. STEMCELL is pleased to offer one $500USD travel award to attend the SickKids’ Cystic Fibrosis training workshop. Winners cannot be funded by any other travel awards, including those offered by CFIT or OIRM. Winners must provide proof of registration and an invoice for travel costs. To apply for this travel award, please fill out the online form. Submissions will be accepted until March 31, 2019. The winner will be announced shortly thereafter.
MBW Training Course
As part of an International effort to standardize the multiple breath washout (MBW) test, the North American MBW training centre at The Hospital for Sick Children offers an intensive MBW Training course and qualification process. The training course is designed to facilitate making real-time decisions about device operation and test quality and is delivered through a comprehensive one day hands-on session combined with two interactive online courses. MBW training information can be found here.
Other courses under consideration for future development:
Nasal cell culture and expansion. (possible future development; $ TBD)
Practical training on culturing primary nasal cells from CF donors and expanding for functional studies. Must have basic laboratory and tissue culture experience. Course is under consideration for a future offering.

