Medulloblastoma
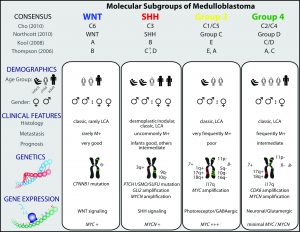
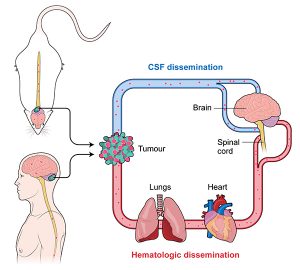
Our Medulloblastoma (MB) projects are driven by the tumour and sequencing data that form part of the Medulloblastoma Advanced Genomics International Consortium (MAGIC). MAGIC is a SickKids-based consortium of clinicians, scientists, and pathologists from over 85 cities around the world who have collectively contributed frozen medulloblastoma samples to the genomics efforts in Toronto.