Replication Repair Deficiency Research
The International Replication Repair Consortium (IRRDC) is an international collaborative research initiative run by our group which aims to better understand and manage this rare genetic condition.
Through collecting biological and clinical data of affected individuals and family members, molecular genetic analysis, patient screening and laboratory research, we continue to increase our understanding of this complex genetic syndrome. Since the establishment of the Consortium, the number of Consortium members is growing, and now includes clinicians, scientists, genetic counsellors, medical professionals, psychologists and patient advocates.
Our Research Initiatives Include:
Clinical Research
Defining the spectrum of CMMRD cancers and their outcome is an ongoing effort. In recent years, important papers from the European group (C4CMMRD, J Med Genet 2014), the French group (Lavoine et al. J Med genet 2015) and the international CMMRD consortium (Backry et al, EJC 2014) identify malignant gliomas, haematological malignancies and gastrointestinal cancers as the most common cancers in CMMRD.
Ongoing research and data collection revealed other less common cancers in children, and common adult cancers such as breast cancers and genitourinary malignancies in adult survivors of CMMRD. These findings have specific implications on the surveillance protocol (Tabori et al. Clin Cancer Res. 2017).
Although the prevalence of CMMRD is unknown, we’ve found that in some areas of the world, such as the Middle-East and South Asia, CMMRD can be common and present cancers such as malignant brain tumors (Amayiri et al. IJC 2015).
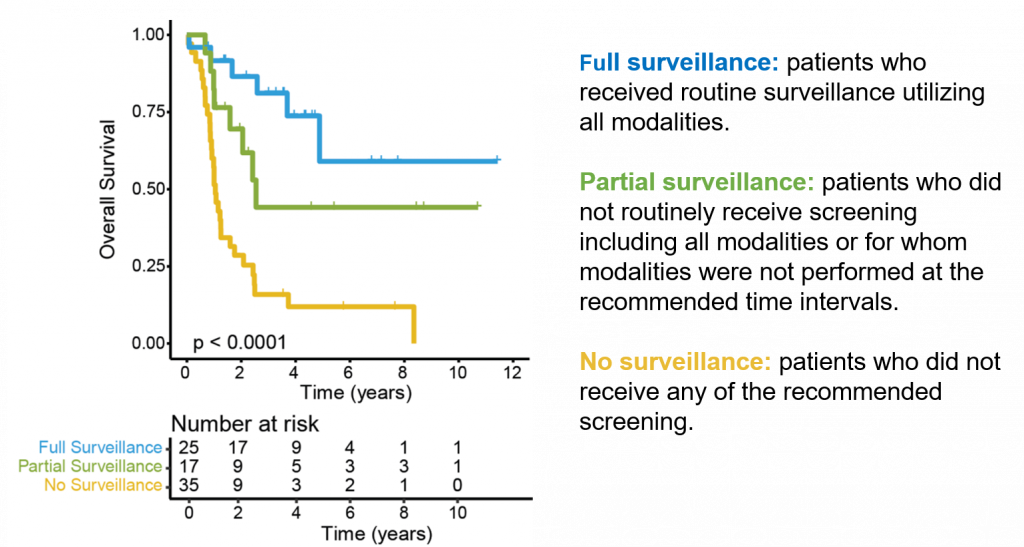
Establishing Effective and Feasible Surveillance Programs
CMMRD patients are at high risk for multiple malignancies throughout childhood. Therefore, a screening protocol was developed and implemented (Tabori et al. Clin Cancer Res. 2017) with the goal of intervening at an early tumor stage. Recent data suggests that CMMRD patients that undergo screening experience improved outcomes.
Through the screening protocol, CMMRD patients are reaching adulthood for the first time.
We‘re monitoring and collecting surveillance data of CMMRD patients to ensure the long term follow up for those patients and families, and to further refine surveillance strategies to improve clinical outcomes and quality of life of our patients and families.
New Diagnostic Approaches
CMMRD is diagnosed by identifying a germline mutation on both alleles of one of four mismatch repair genes (MSH2, MSH6, MLH1, PMS2). But clinical sequencing can be costly and problematic, particularly in the case of PMS2, for which many pseudo genes exist that interfere with the correct identification of a mutation.
We’ve shown that microsatellite instability (MSI) is not a reliable predictor of CMMRD, as it is in Lynch syndrome. Therefore, the Consortium has been addressing the need for accurate and sensitive diagnostic tools since its conception.
The use of more accessible and highly sensitive diagnostic tools have been tested and validated by the Consortium and others. New diagnostic tools include immunohistochemistry staining for the four MMR genes in both normal and tumor tissue, functional tests that measure a patient’s ability to repair mismatches, and a patient’s cell line response to chemotherapeutics that require MMR function. These tools can be implemented from a simple blood draw or skin biopsy, and don’t incur the cost of traditional sequencing.
Basic and Translational Research
Using normal and tumor tissues from members of the Consortium, we found that CMMRD cancers have the highest mutational load among all human cancers (Shlien et al, Nature Genet 2015). We’ve also uncovered secondary somatic mutations in genes such as DNA polymerase E and D which cause this hypermutant phenotype. Current research is focused on ways to use this phenomenon as a weakness of these cancers and to develop new therapies aiming at hypermutation.
The Consortium is collaborating to study other aspects of CMMRD tumors, such as the effect of hypermutation on RNA and on DNA methylation.
We’re developing animal models to study the mechanisms of cancer initiation and progression in CMMRD. We also use patient–derived xenograft tumors to test new drug therapies as preclinical models before moving to clinical trials.
Modeling DNA Replication Repair Deficiency in Zebrafish
One of the models we’re using is zebrafish, a tropical fish growing in popularity as a scientific model due to its fast-breeding time, transparency at its embryonic and larval stages, and the conservation between humans and zebrafish in disease-related genes.
The modeling of DNA replication repair deficiency in zebrafish has two objectives:
- To model the human CMMRD-driven cancers that lack replication repair
- To conduct a high-throughput drug screen to identify potential therapeutics that can overcome the intrinsic drug resistance of cells that do not have replication repair
Modeling DNA Replication Repair Deficiency in Mice
The absence of animal models of CMMRD brain tumors is a barrier for rapid testing of drug efficacy.
We‘ve designed and characterized new mouse models of these malignant brain tumors. Such models have been crucial to further our understanding of biological mechanisms driving CMMRD cancers.
Immunotherapy—Checkpoint Blockade
Immunotherapies targeting and inhibiting immune checkpoints have recently shown efficacy in individuals with metastatic melanoma, non-small cell lung carcinoma (NSCLC) and mismatch repair deficient colorectal cancers (Lynch Syndrome).
The underlying biology connecting these cancers is their high rates of mutation. We now know that this increased mutation load causes these cancers to display higher numbers of neoantigens. This means that the tumors are more likely to be detected as “non-self” and thus be targeted and attacked by the body’s immune system. Since CMMRD brain tumors harbor the highest mutation load of all paediatric cancers, as well as most adult cancers, this makes them susceptible to immunotherapies such as Nivolumab.
Our group is currently working to characterize the immune response triggered by this treatment through new tools to gain better knowledge and identify key immune factors responsible for the response. This allows us to determine how and if a patient will benefit from immunotherapy. We’re also investigating our immunotherapy functions when combined with other novel agents.
Clinical Trials
Based on our translational data, we recently observed encouraging response to a new type of immunotherapy called immune checkpoint inhibitors. Recurrent glioblastoma from CMMRD patients with hypermutation responded to the immune checkpoint inhibitor Nivolumab (Bouffet et al. JCO 2016).
In order to test new treatments for CMMRD, the International CMMRD Consortium is supporting two new international clinical trials which will test responses to immune checkpoint inhibition in recurrent CMMRD cancers. Biological correlative studies will be performed to determine the reasons for response or lack of for these therapies.
Replication Repair Deficiency Team
- Sumedha Sudhaman, PhD
Research Fellow
- Owen Crump
Research Technologist I
- Victoria Forster, PhD
Research Fellow
- Logine Negm
M.Sc. Candidate
- Cindy Zhang
Lab Manager
- Ayse Bahar Ercan
M.Sc. Candidate
- Melissa Galati , PhD
PhD Candidate
- Nuno Miguel Nunes, PhD
Research Fellow
- Melissa Edwards, PhD
Clinical Research Project Manager
- Jiil (Brian) Chung
PhD Candidate










